Price negotiations underway for Zydus Cadilla Vaccines
Zydus Cadilla Vaccines related price negotiations going on…
Tuesday, 5th October 2021
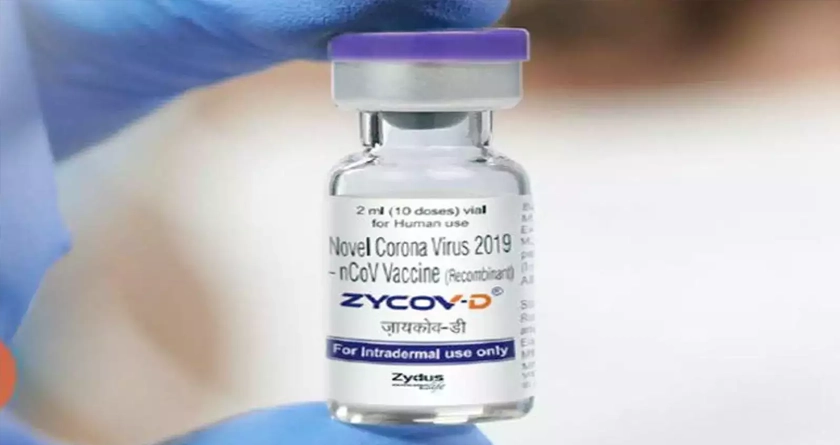
As Ahmedabad based pharmaceutical company Zydus Cadilla gears up to roll out the world’s first DNA based “ZyCov-D” vaccines, negotiations over the pricing for its procurement remains under deliberation with the Centre and is likely to reach a conclusive decision this week.
The government as on Thursday released a statement announcing ZyCov-D vaccines to be introduced nationwide as a part of Covid immunization drive.
According to sources, the pharma company has put forth a price quotation of Rs 1900/- inclusive of all taxes for its three dose needle free regimen to be administered for age groups of 12 years and above. Based on an official source, the ministry is still awaiting National Technical Advisory Group (NTAGI) directives to introduce ZyCov-D as a part of the inoculation drive to be applied for the majority aged 12 to 18 with underlying health issues.
Meanwhile the government’s initiative to reduce the costs of the jabs is still underway and is expected to come to a final decision this week. The pharma company is asked to reconsider its prices of the vaccines in an attempt to make it more affordable for the masses. Sources reveal that as far as around three rounds of discussions have taken place between the Centre and the company to negotiate the terms of pricing.
During a press conference on Thursday, Union Health Secretary Rajesh Bhushan gave the statement that the government is in discussion with the manufacturers. He concluded by saying that as this is a three-dose vaccine that comes with a needleless delivery system, the terms for its pricing is likely to be different than the existing vaccines for Covid immunization programme.
Unlike its contemporaries Covishield, Sputnik-V and Covaxin vaccines are administered to masses only above 18 years of age, while Zydus Cadilla’s “ZyCov-D” vaccine is the first to receive Emergency Use Authorization from the drug regulator on August 20 to be administered to children of 12 to 18 years of age.
Developed in coalition with the department of biotechnology, ZyCov-D is first of its kind vaccine based on plasmid DNA technology that generates a spike of SARS COV-2 to trigger an immune response. A needle less vaccine that will be administered intradermal, i.e. on the skin to potentially mitigate the side effects.
The News Talkie Bureau
Source- Business-Standard